The steam distillation is a process in which the concentration and isolation of essential oil from reliable and often cheaper natural sources is carried out. This is an important technique and has significant commercial applications. Many compounds, both solids, and liquids are separated from complex mixtures by taking advantage of their volatility in steam. A compound must satisfy three conditions to be successfully separated by steam distillation. It must be stable and relatively insoluble in boiling water and must have a vapor pressure in boiling water that is of the order of 1 kPa (0.01) atmosphere. If two or more compounds satisfy these three conditions, they will generally not separate from each other but will be separated from everything else.
Table of Contents
Principle of Steam distillation
The principle behind steam distillation is a way of separating miscible liquid based on their volatilities. The boiling point of the products is so minimized that it permits the constituents to get vaporized. The vapor pressure exerted by the liquids differs in strength, which is a function of temperature. The boiling of the liquids takes place and at a certain instance of time, the boiling point of the natural products in the liquid form surpasses the atmospheric pressure. The result is that the vapor pressure of the whole system increases.
Construction of Steam distillation
A large-scale stainless steel steam distillation unit usually has a capacity from 0.5 to 15 thousand liters. Its usual diameter varies from 1.5 to 5 meter. The still contains jacket through which steam can be passed for heating the content, Fig. 6.8. Steam vessel is hydrostatically tested at 125 psi to serve as the distillation tank. Low pressure or high-pressure steam is supplied by a boiler. The steam vessel can hold extract approximately 3 to 5 liters of essential oil per distillation. The size of the tub is designed to provide oil in sufficient quantity for industrial evacuation or for analysis. The vapors of water and volatile oil are condensed in the condenser attached. The distillate is collected as two layers which are separated by a Florentine receiver. Following the distillation, the vessel can be disconnected from the cold-water condenser and rotated on swivels to a horizontal position, permitting easy removal and refilling of plant material. The entire extraction unit (vessel, condenser, boiler, and oil collector) is suitable for mounting and transportation. It is built to extract volatile essential oils from aromatic plants.
Working of Steam distillation
Steam distillation is a process employed to extract essential oils from organic plant matter by passing steam generated through the plant material. Usually, a chamber is filled with holes (perforations) in the bottom for steam to come through with either fresh or dried herbs.
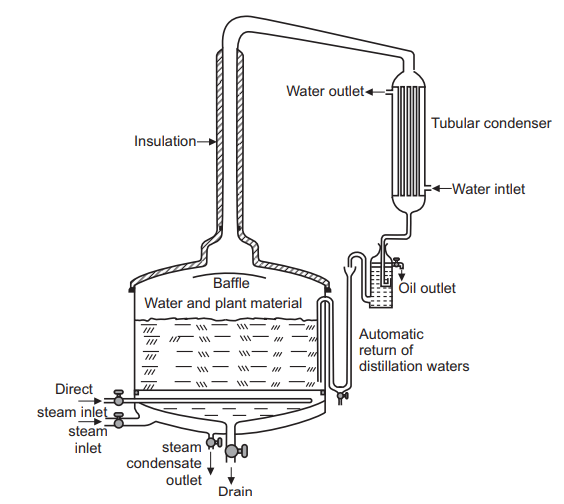
Temperature-sensitive compounds which would normally decompose through simple distillation vaporize at lower temperatures when subjected to steam in the distillation column. This allows for the separation of essential oils, which tend to be less soluble in boiling water, from chemically complex materials. When the steam is passed through the organic material, tiny pockets holding the essential oils open to release the essential oil molecules without doing any damage to these delicate components. A lid keeps the oil from diffusing into the air when the steam is applied to it. The distillate obtained contains a mixture of water vapor and essential oils which returns to their liquid form in the condensing apparatus. The condensed water and oil droplets are collected and channeled them through a filter, which separates the water from the oil. They are separated using a Florentine separator. In the case of essential oils, using steam allows the distillate to retain the more delicate flavours and aromas that would otherwise break down if high temperatures are applied.
Applications of Steam distillation
(i) Steam distillation is used to extract essential oils from aromatic plants to flavour liqueurs.
(ii) It is used at a wide scale for the manufacturing of essential oils like perfumes.
(iii) It is used in the synthesis of complex organic compounds.
(iv) Orange oil and eucalyptus oil are obtained at an industrial scale using this method.
(v) It is also used in petroleum industries and in the production of consumer food products.
(vi) It is used for the extraction of peppermint and spearmint oils.
Advantages
(i) Steam distillation is useful for extracting most fats, oils, and waxes. This process works well for types of substances that do not mix with water.
(ii) It can be a cost-effective method to invest in to extract a diverse array of immiscible substances.
(iii) Since steam temperature can remain at the boiling point of water; this process also has a cost-benefit of requiring less fuel for the steam boiler.
(iv) The amount of steam and the quality of the steam can be controlled.
(v) Lower risk of thermal degradation as temperature generally does not rise above 100 °C.
(vi) Most widely used process for the extraction of essential oils on a large scale.
(vii) It is the standard method of extracting flavour and fragrance.
Disadvantages
(i) Need trained operator in order to operate the equipment.
(ii) The process has a hidden cost of maintaining and repairing equipment.
(iii) There is a much higher capital requirement and with low-priced oils the payback period can be over 10 years.
(iv) Requires a higher level of technical skill and fabrication and repairs and maintenance require a higher level of skill.