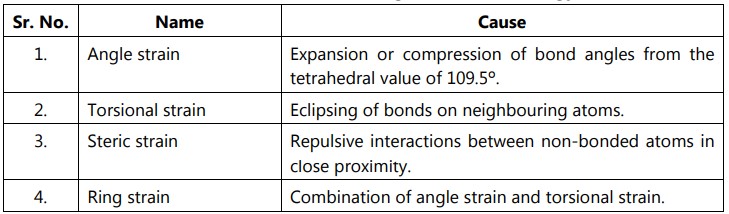
Conformational isomerism: These are the isomers formed when single bonds in the molecule are rotated. Through additional rotation around single bonds, they might be quickly interconverted back to one another. One conformer must be transformed into another by overcoming a rotating energy barrier.
Some important examples of conformational isomerism include:
(i) Open chain alkane conformations: Staggered, eclipsed, and gauche conformers.
(ii) Ring conformation: Chair and boat conformers.
(iii) Atropisomerism: A molecule can become chiral due to restricted rotation about a bond.
(iv) Folding of the molecule.
Table of Contents
Conformations of Ethane-Conformational isomerism
Out of infinite conformations possible, most important conformers of ethane are:
(i) Staggered conformation and (ii) Eclipsed conformation.
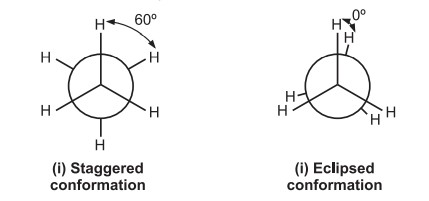
The staggered conformation of ethane is the most stable shape because the angle between the C-H bonds of the first and second carbons is 60o. When two C-H bonds are eclipsed, their angle is 0°, which causes repulsion in their electron clouds, increasing the molecule’s energy and decreasing its stability. Ethane has a 3 kcal/mol worse stability in its eclipsed structure compared to its staggered conformation.
The bulky substituents of the molecule are pulled closer together in the eclipsed conformation, which causes them to repel one another. This obstruction makes spinning more difficult (torsional strain i.e., the force that opposes rotation due to the repulsion of bonding electrons.) Due to their quick interconversion at ambient temperature, neither of the ethane conformations can be isolated.
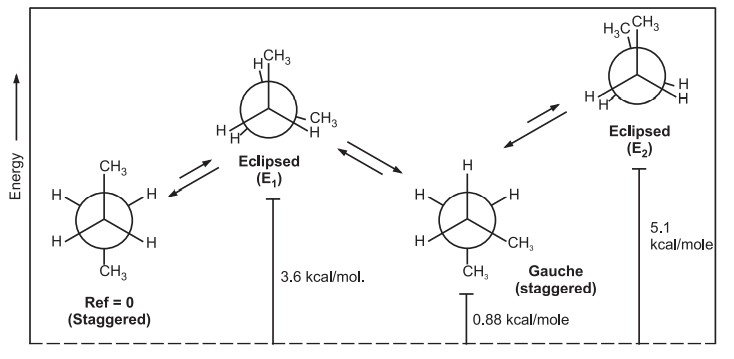
Conformations of n-Butane–Conformational isomerism
Various conformations of n-Butane include,
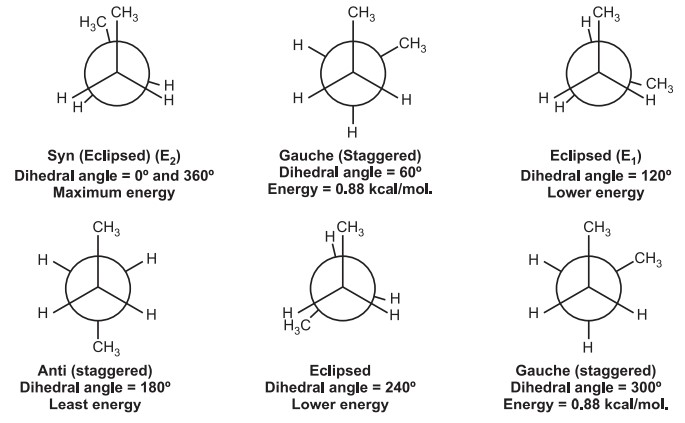
Dihedral angle: It is the angle created by two intersecting planes.
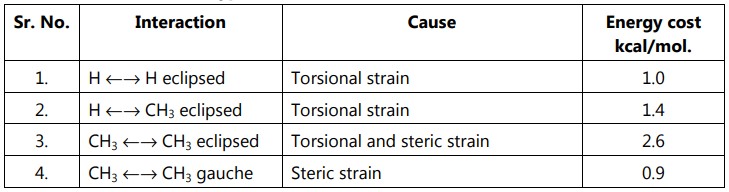
The gauche butane is less stable than antibutane by 3.8 kJ/mol. because of steric interference between H-atoms on the two methyl groups.
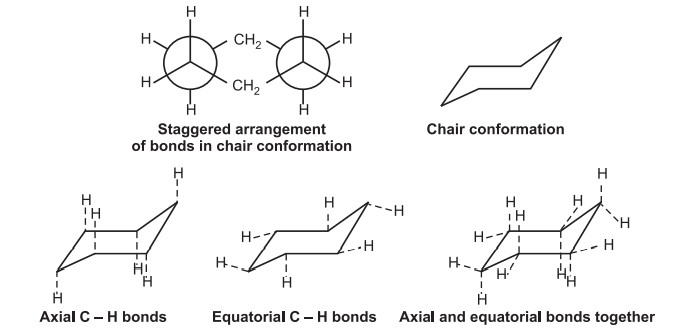
Conformations of Cyclohexane–Conformational isomerism
In cyclohexane, all carbon atoms are sp3 hybridized with a bond angle of 109º. This leads to two types of conformations.
(i) Chair conformation:
It is the most stable form having a tetrahedron bond angle of 109º. It adopts a staggered arrangement having the least torsional strain.
Chair cyclohexane has six axial hydrogens perpendicular to the ring (parallel to the ring axis) and six equatorial hydrogens near the plane of the ring.
Six membered rings are almost free of strain in a chair conformation.
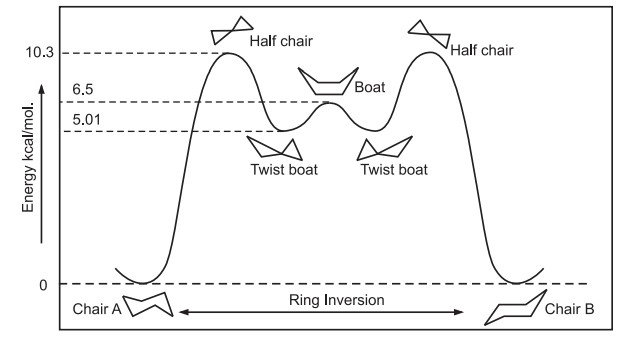
By bending the links, the chair conformation can be converted into a boat form. Through intermittent half-chair and twist boat forms, the chair can change into a boat. While all hydrogens in the chair conformation are staggered, four hydrogens are eclipsed in the boat conformation, where carbon 1 and carbon 4 are bent toward one another. Consequently, the boat conformation is 6.5 kcal/mol less stable than a chair conformation.
Chair conformations easily interconvert, changing the axial and equatorial orientations as a result of the simultaneous rotation about all C-C bonds. It is referred to as a ring flip or inversion. As a result of this transformation, axial bonds turn into equatorial bonds.
Atropisomerism: A chiral axis is produced by restricted bond rotation, which results in atropisomerism, which is stereochemistry. Atropisomers are stereoisomers formed by impeded rotation at one or more single bonds between two planar moieties when the energy barrier to rotation is sufficiently high to permit the separation of individual conformers. If the conformers’ half lifetimes are greater than 10-2 seconds, NMR can detect them, and if they are greater than 1000 seconds, isolation is possible.
Although Christie discovered atropisomerism in 6,6-dinitro-2, 2′-diphenic acid in 1922, Kuhn initially coined the term in 1933. Atropisomerism is derived from the Greek words a = not and tropos = turn. At normal temperature, the larger groups on the ortho position of the biphenyl ring prevent the rotation across the C-C bond, giving rise to two enantiomers.
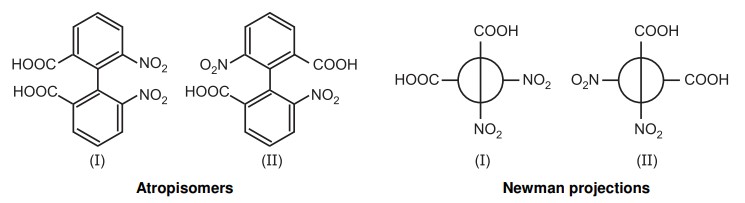
Atropisomerism induces time-dependent inversion of chirality via bond rotation generating atropisomers having different pharmacokinetic, biological, and toxicological profiles.
Also read: Determination of configuration of geometrical isomerism