Conditions for optical activity : The two aryl groups’ planes must not be plane. Bulky groups are positioned in the ortho locations to achieve it.
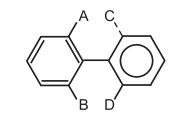
(ii) In most cases, the enantiomers can be resolved.
(iii) Ortho substituents increase the restricted rotation by their steric repulsion.
(iv) Mono ortho-substituted biaryl compounds do not show atropisomerism at room temperature. e.g.
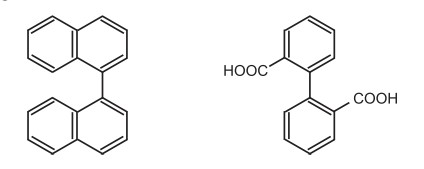
v) In addition to the substitutents at the ortho position, the bulky groups adjacent to the ortho substitutents increase the stability and isolatability of atropisomers.
(vi) Heteroaromatic system provides chirality even though their ortho substitute they are same.
Also read: Determination of configuration of geometrical isomerism