The basic idea behind fractional distillation is the same as simple distillation. The difference between simple and fractional distillation is the number of times that the liquid is vaporized and condensed. Simple distillation condenses the liquid once, so the boiling points of the two liquids must be far apart to make it efficient. Simple distillation is performed on a mixture of liquids with similar volatilities and the resulting distillate is in a more concentrated form in the more volatile compound than the original mixture and it may contain a significant amount of the higher boiling compound. If the distillate of simple distillation is distilled again, the resulting distillate is again of further more concentrated form of the lower boiling compound, but still, a portion of the distillate contains the higher boiling compound. The number of simple distillations in a fractional distillation depends on the length and efficiency of the fractionating column.
Fractional distillation is a process in which vaporization of a liquid mixture gives rise to a mixture of constituents from which the desired one is separated in pure form. This method is also known as rectification because a part of the vapour is condensed and returned as a liquid. This method is used to separate miscible volatile liquids, whose boiling points are close, by means of a fractionating column. Fractional distillation is different from simple distillation. In simple distillation, the vapour is directly passed through the condenser. In fractional distillation, the vapour must pass through a fractionating column in which partial condensation of vapour is allowed to occur. In simple distillation, condensate is collected directly into the receiver, while in fractional distillation condensation takes place in the fractionating column so that a part of the condensing vapour returns to the still.
Table of Contents
Principle of fractional distillation
When a liquid mixture is distilled, the partial condensation of the vapour is allowed to occur in a fractionating column. In the column, ascending vapours from the still are allowed to come in contact with the condensing vapour returning to the still. This result is the enrichment of the vapours with the more volatile component. By condensing the vapour and reheating the liquid repeatedly, equilibrium between liquid and vapour is set up at each stage, which ultimately results in the separation of a more volatile component.
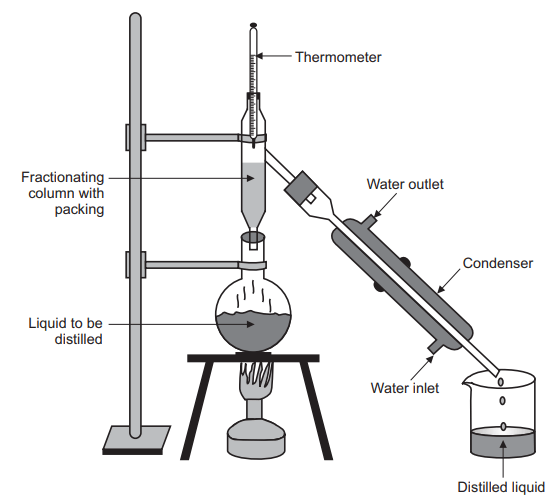
Construction of fractional distillation
The equipment used for fractional distillation consists of a special types of still-heads known as fractionating columns. In still-heads, condensation and revaporisation are affected continuously. Fractionating column is essentially a long vertical tube in which the vapour moves upward and partially gets condensed. The condensate flows down the column and is returned to the flask. The columns are constructed to provide a large cooling surface for the vapour to condense and to obstruct the ascending vapour to allow easy condensation. The obstruction also retards the downward flow of liquid, which has a high boiling component. The fractionating columns used are packed columns and plate columns.
Packed columns
In this column, some form of packing is used to affect the necessary liquid/vapour contact. The packing consist of single turn helices (spirals) of wire or glass, glass rings, cylindrical glass beads, stainless steel rings etc. The column consists of a tower containing a packing that becomes wetted with a film of liquid, which is brought into contact with the vapour in the intervening spaces. The same type of fractionating columns can be obtained in various lengths. A long fractionating column is necessary when the boiling points of the constituents are lying fairly close together. A short fractionating column is necessary when the boiling point of the constituents differ considerably. Packing must be uniform so as to obtain proper channels. If packing is irregular, the mass transfer becomes less effective. An example is the Widmer column.
Plate columns
Many forms of plates are used in the distillation columns. These can be divided into Bubble cap plates and Turbo grid plates. The bubble cap column is used in large distillation plants. The column consists of a number of plates mounted one above the other. Caps are present on each plate, which allow the vapour to escape by bubbling through the liquid. Ascending vapour from the still passes through the bubble-caps on plate A and the rising vapour will be richer in the more volatile component. This vapour passes through the liquid on plate B and is partially condensed. The heat of condensation partially vaporizes the liquid. The process of condensation and vaporization is repeated at plate C and so on all the way up the column. Each bubble-cap plate has the same effect as a separate still. The bubble cap plate is effective over a wide range of vapour-liquid proportions. There is an excellent contact as the vapour bubbles through the liquid. The major limitation is a layer of liquid on each plate that results in considerable hold-up of liquid over the entire column. There is a need to force the vapour out of the caps, through the liquid that leads to a large pressure drop through the column. In addition, the column does not drain when it is not in use. The structure of bubble plate is so complicated that makes construction and maintenance expensive.
Working of fractional distillation
In fractional distillation, the vapours formed from the boiling mixture rise into the fractionating column where they condense on the column’s packing. This condensation is similar to a single run of simple distillation; the condensate is more concentrated in the lower boiling compound than the mixture in the distillation flask. As vapors continue to rise through the column, the condensed liquid revaporizes. Each time this occurs the resulting vapours are more and more concentrated in the more volatile substances. The length of the fractionating column and the material it is packed with the impact the number of times the vapours will recondense before passing into the condenser. The number of times the column will support this is referred to as the number of theoretical plates of the column. The procedures of simple distillation are so similar to those involved in fractional distillation; the apparatus that is used in the procedures are also very similar. In fractional distillation, a packed fractionating column is attached to the top of the distillation flask and beneath the condenser. This provides the surface area on which rising vapours condense, and subsequently revaporize.
The fractionating column is used to supply a temperature gradient over which the distillation can occur. In an ideal situation, the temperature in the distillation flask would be equal to the boiling point of the mixture of liquids and the temperature at the top of the fractionating column would be equal to the boiling point of the lower boiling compound; all of the lower boiling compound would be distilled away before any of the higher boiling compound. In reality, fractions of the distillate must be collected because as the distillation proceeds, the concentration of the higher boiling compound in the distillate being collected steadily increases. Fractions of the distillate, which are collected over a small temperature range, will be essentially purified; several fractions should be collected as the temperature changes and these portions of the distillate should be distilled again to amplify the purification that has already occurred.
Efficiency of Fractional distillation
The efficiency of separation by fractional distillation of a mixture may depend upon various factors that include fractionating column, reflux ratio, heat input and column temperature etc. The reflux ratio is the quotient of the amount of liquid returning through the column to the amount collected into the receiver during the same interval of time. A column that operates under total reflux will not yield distillate and thus it should be high. It is controlled by selecting proper still. Other experimental conditions necessary for good separation are a comparatively large amount of liquid continuously returning through the column, thorough mixing of liquid and vapour and a large active surface of contact between liquid and vapour. The number of vaporization-condensation cycles that can occur within a fractionation column determines the purity which can be attained. The efficiency of a column depends upon column length and composition. Generally, the column is packed with a copper sponge. This increases the surface area that the ascending vapour encounters and results in more vaporization-condensation cycles compared to an empty column.
Theoretical Plates
A measure of the efficiency of a column is known as the number of theoretical plates of that column. One theoretical plate is equivalent to one vaporization[1]condensation cycle which is equivalent to the one simple distillation. Thus a fractionation column that can attain the equivalent of three simple distillations would be said to have three theoretical plates.
Applications of fractional distillation
(i) Fractional distillation is used for the separation of miscible liquids such as acetone and water, chloroform and benzene.
(ii) Fractional distillation is suitable for a system when the boiling point of the mixture is always intermediate between those of pure components.
(iii) There is neither a maximum nor a minimum in the composition curves (azeotropic mixtures). Examples include benzene and toluene, carbon tetrachloride and cyclohexane, and water and methanol.
Advantages
(i) Frictional distillation gives good solvent recovery
(ii) Fractional distillation is easy to use and operate.
(iii) Fractional distillation is also highly efficient, especially for systems that use stacked distillation columns, which produce more output at lower costs.
(iv) It helps to produce much-needed fuel.
Disadvantage
(i) Fractional distillation cannot be used to separate miscible liquids, which form pure azeotropic mixtures.
(ii) It is expensive because it requires large structures, heavy-duty materials, and specialized machinery.
(iii) It also requires staff to be fully trained in the operation of systems to ensure they know how to use the distillation equipment and won’t make mistakes.
(iv) It presents a wide range of risks for the people who are involved in it. For example, explosion.
(v) It can contribute to environmental pollution. Fractional distillation in itself is not harmful but the types of mixtures that are distilled disturbs ecology. For example, refining crude oil.