Electron arrangements in orbits: Having known that planetary electrons numerically equal to the atomic number are revolving about the atomic nucleus in closed orbits, the question arises as to how they are arranged in these orbits.
Langmuir Scheme
Langmuir deserves credit for proposing the first detailed system for the organization of extranuclear electrons in 1919. His basic premise is that inert gases have the most stable electron configuration and, as a result, include full electron orbits. The initial orbit of helium is considered fully saturated with two electrons since it possesses two planetary electrons.
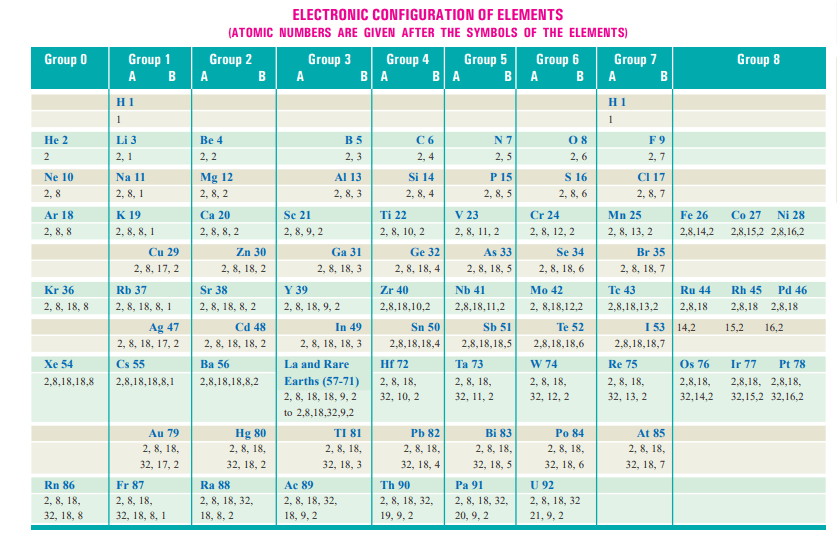
We have 10 planetary electrons in the next inert gas neon, and since two of them would fully saturate the first orbit, the remaining eight will create the next stable orbit. Argon, which has an atomic number of 18, will have the same arrangement of 2, 8, 8. The subsequent orbits would include 2, 8, 8, 18, 32 electrons if we continued in this fashion. Although true for the first few components, Langmuir’s approach fails to explain the behavior of higher elements.
Bohr-Bury Scheme
Bury proposed a version of the Langmuir method in 1921 that he said was more in line with the physical and chemical characteristics of specific elements. At about the same time as Bury developed his scheme on chemical grounds, Bohr (1921) published independently an almost identical scheme of the arrangement of extra-nuclear electrons. He based his conclusions on a study of the emission spectra of the elements. Bohr-Bury scheme as it may be called can be summarised as follows :
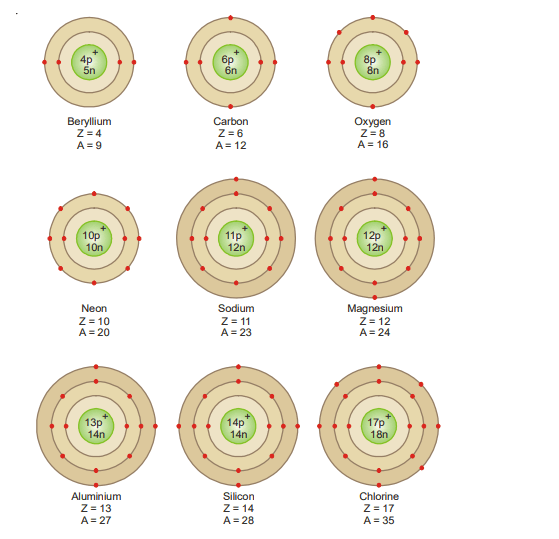
Rule 1. The maximum number of electrons that each orbit can contain is 2 × n2, where n is the number of orbits.
The first orbit can contain 2 × 12 = 2 ; second 2 × 22 = 8 ; third 2 × 32 = 18 ; fourth 2 × 42 = 32, and so on.
Rule 2. The maximum number of electrons in the outermost orbit is 8 and in the next-to-the outermost 18.
Rule 3. It is not necessary for an orbit to be completed before another commences to be formed. In fact, a new orbit begins when the outermost orbit attains 8 electrons.
Rule 4. The outermost orbit cannot have more than 2 electrons and the next-to-outermost cannot have more than eight so long as the next inner orbit, in each case, has not received the maximum electrons as required by rule (1).
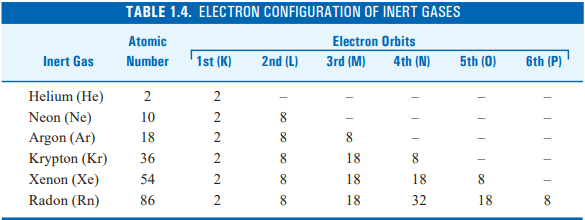
According to the Bohr-Bury scheme the configuration of the inert gases is given in the table below :
A complete statement of the electron configuration of elements elucidating the various postulates of the Bohr-Bury scheme is given in the table on page 31 for ready reference.
Make sure to check our amazing article: Sommerfeld’s modification of bohr atom